The following CDRC resources are available to support safe Corticosteroid/ Steroid Prescribing.
Steroid Template
How to Access:
In the lower left hand corner use the search bar, type in ‘Steroids – CDRC’ and select the following template:

Alternatively, press F12 and search for ‘Steroids – CDRC’, this will open the aforementioned template.
Steroids – CDRC Template
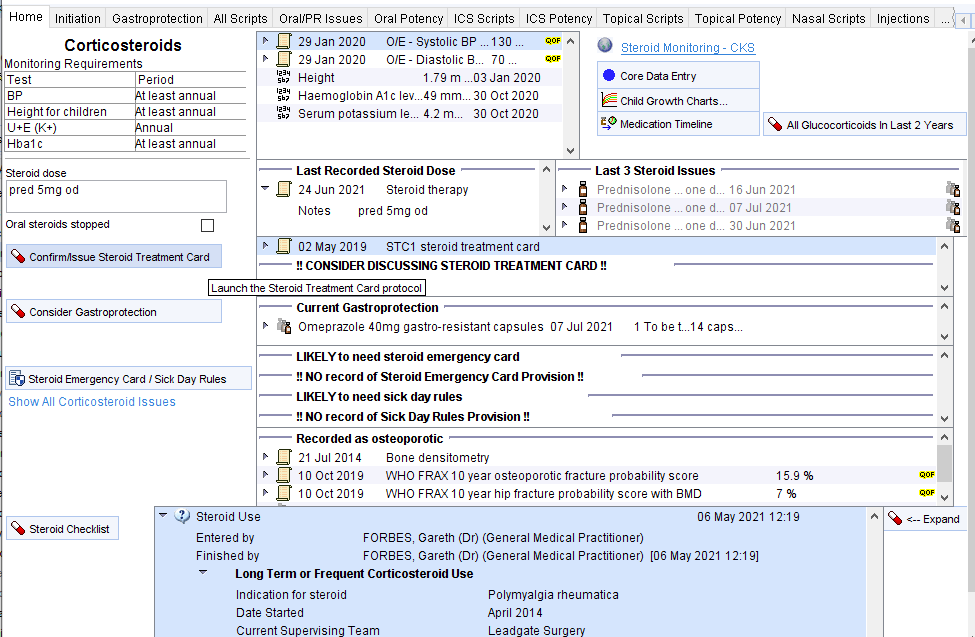
This template has three columns:
| Data Entry / Links to other templates/tools | Relevant information from the record | Links to expanded information |
| Monitoring requirements for oral steroids | Most recent information about monitoring | Link to CKS guidance on monitoring Link to Core Data template to record height/weight etc. Link to child growth charts Medication Timeline Link to view showing all steroid issues in the last 2 years (all forms – e.g., oral, topical, nasal etc.) |
| Space to record current oral steroid dose | Most recently recorded oral steroid dose | Last three oral steroid issues – helps to look at concordance, frequency of use |
| Button to record that steroid treatment card has been issued | Most recent record of steroid treatment care issue/discussion Warning will be displayed if STC needs to be considered | |
| Link to Steroid Emergency Card and Sick Day Rules information | Estimation of whether SEC or SDR are needed and whether these have been provided, may need renewal | |
| Information about bone health | ||
| Button to run oral steroid checklist | Latest steroid checklist information | Button to expand the latest information |
Steroid Emergency Cards and Sick Day Rules
This section covers three main areas:
- Identifying people who need SEC or SDR
- Checking Eligibility For Steroid Emergency Card / Sick Day Rules
- Providing Steroid Emergency Card / Sick Day Rules
Identifying People who need Steroid Emergency Cards (SEC) or Sick Day Rules (SDR)
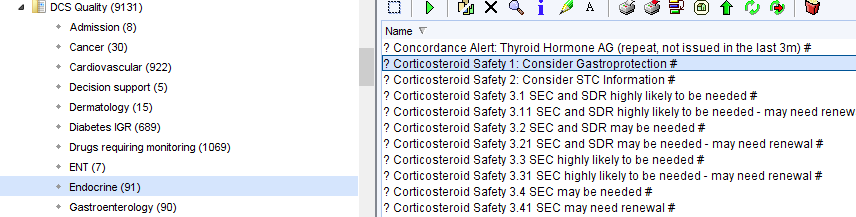
- The following reports, in the CDRC Quality > Endocrine folder, will identify people who may need SEC and/or SDR.
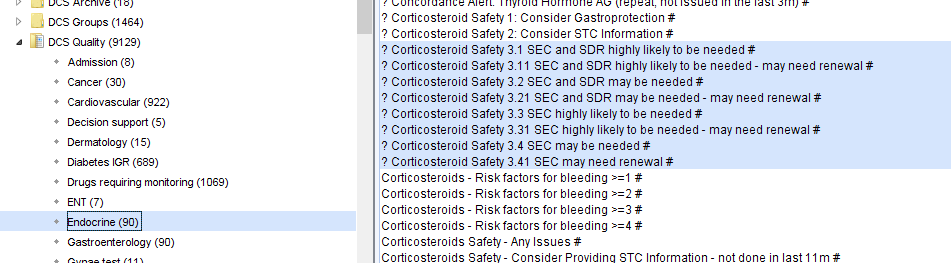
| Report Name | Patients | Action |
| ? Corticosteroid Safety 3.1 | People who are highly likely to need SEC and SDR who haven’t already been given both | Review record and issue as appropriate |
| ? Corticosteroid Safety 3.2 | People who may need SEC and SDR who haven’t already been given both | Review record and check that patient is eligible for SEC and SDR +/- issue them |
| ? Corticosteroid Safety 3.3 | People who are highly likely to need SEC only who haven’t already been one | Review record and issue as appropriate |
| ? Corticosteroid Safety 3.4 | People who may need SEC only who haven’t already been given one | Review record and check that patient is eligible for SEC +/- issue |
The x.x1 searches identify the same people in the searches above who haven’t had a record of SEC/SDR for at least 13 months – consider renewing the information for these patients.
- The Steroid template will show a warning about need for and provision of SEC and SDR.

- The Medication Review Tool template will show a warning. NB. this template will be launched automatically when a medication review is recorded on the repeat template screen (if this has been enabled at your unit).
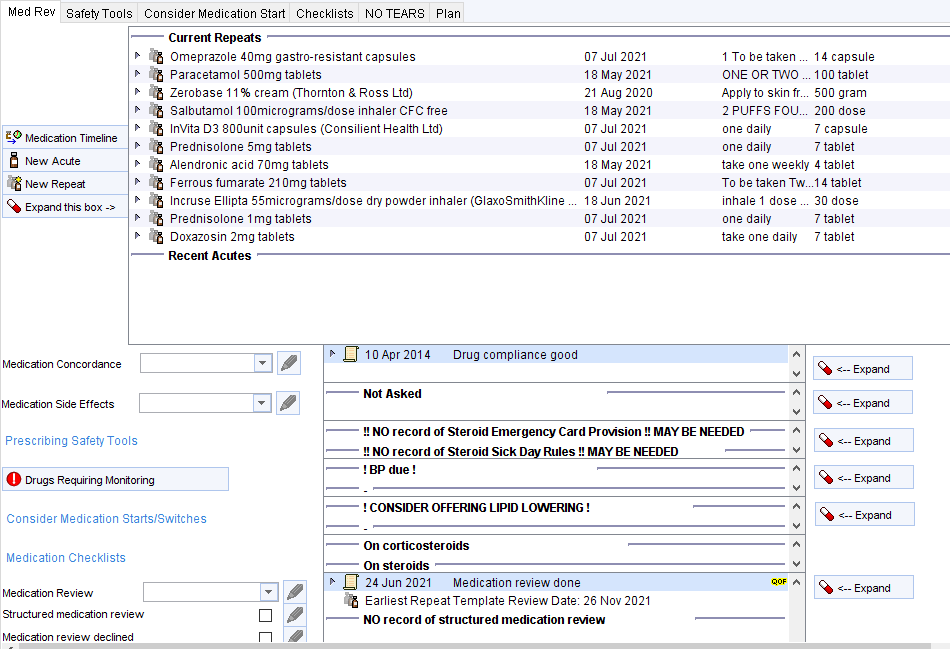
- The LTC Master template will show a warning so SEC/SDR can be discussed during LTC reviews.
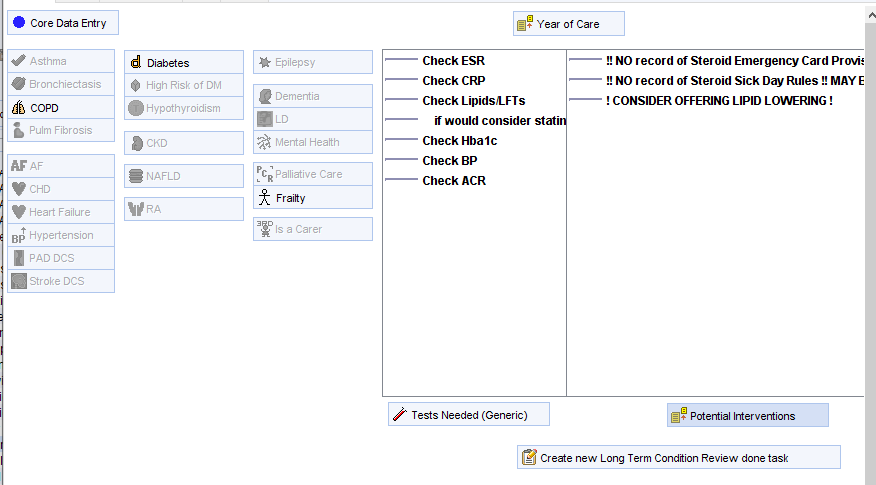
- Relevant LTC review templates will display SEC/SDR warnings – e.g. asthma.

- If a medication is started that suggests the patient may need SEC or SDR a prompt will be displayed.
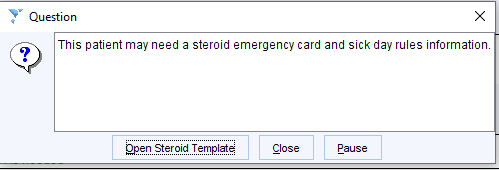
The Steroid template can be opened to check the requirements and to issue the SEC/SDR.
Checking Eligibility for Steroid Emergency Card / Sick Day Rules
The guidance for who needs SEC and SDR is quite complex. The Steroid template will help to check eligibility.
The home page gives an estimate of the requirements.

Click on Show All Corticosteroids to display all corticosteroids issued in the last 2 years.
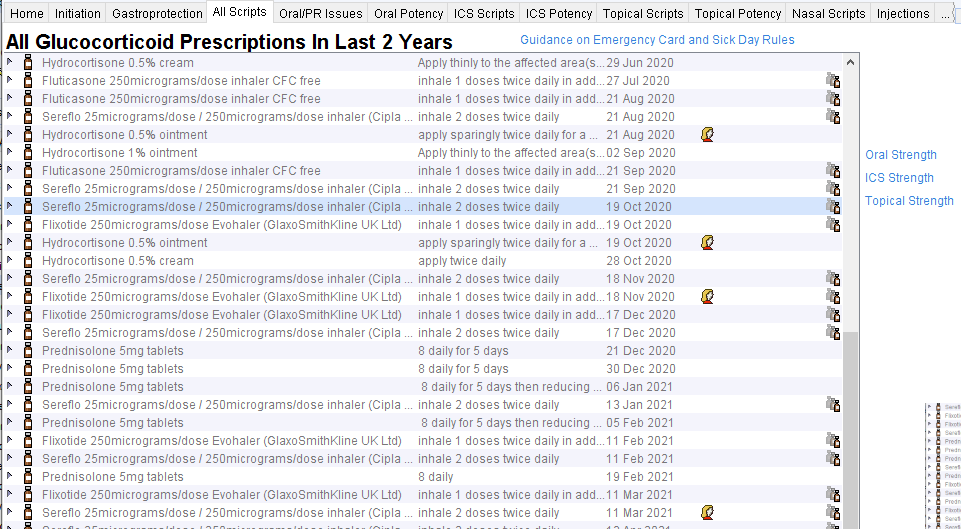
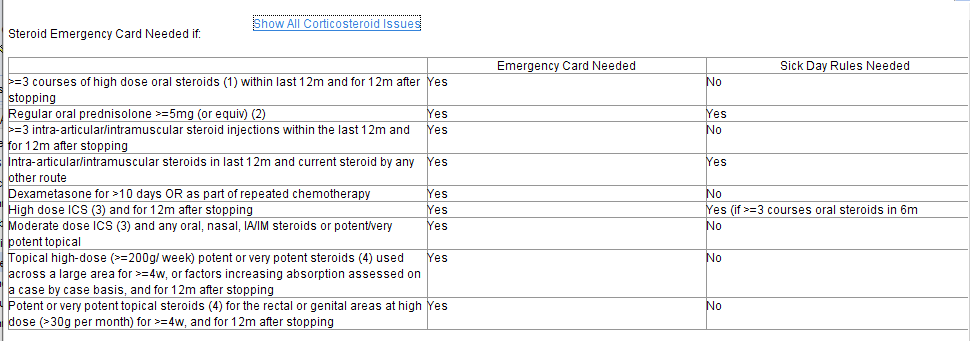
Guidance on Emergency Card and Sick Day Rules links to a page which shows the guidance. The ‘Strength’ links take the user to guidance about relative strengths of different steroid preparations which are needed to interpret the guidance (examples below).

Providing Steroid Emergency Card / Sick Day Rules

Click the Steroid Emergency Card / Sick Day Rules button to open the patient information template.
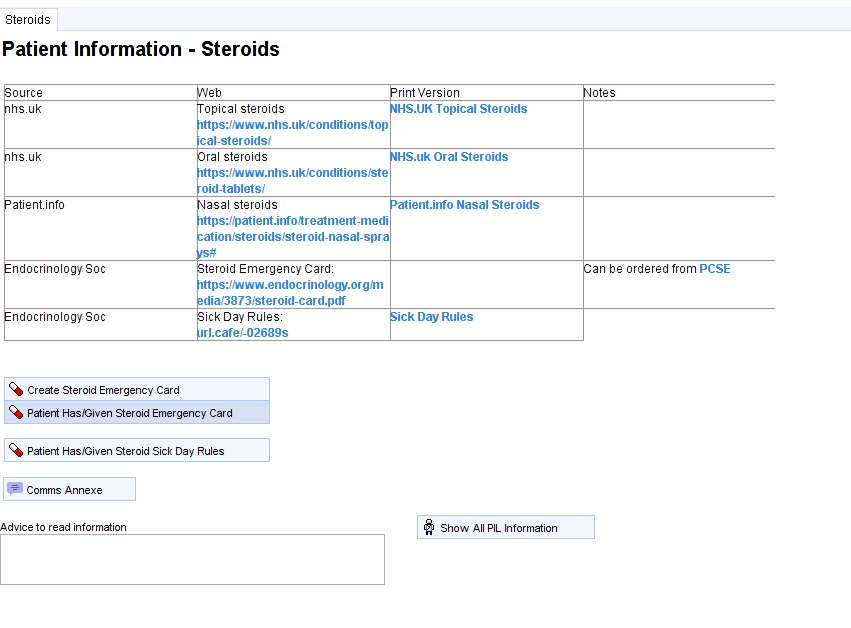
From the table the SEC/SDR can be printed in the surgery or the link can be sent to the patient by SMS/email, using the Communication Annexe
A patient specific SEC can be created within S1 using the Create Steroid Emergency Card button. This will print to be cut out to credit card size (works best with a colour printer).

Once the patient has been provided with the relevant information, click the Patient Has/Given SEC/SDRs buttons as appropriate.
Steroid Monitoring
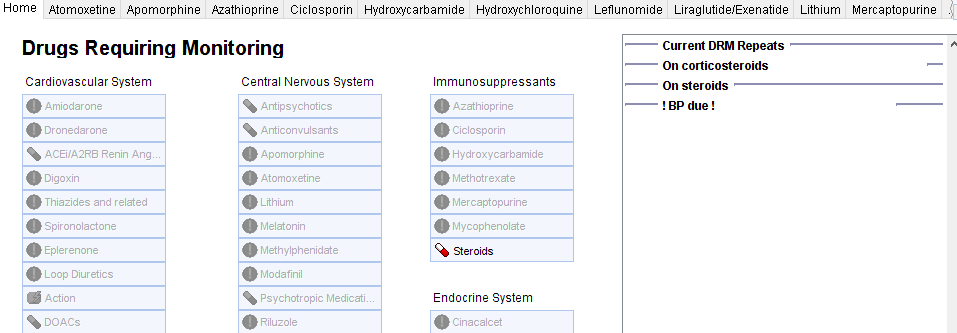
Monitoring of steroids is integrated into the Drug Requiring Monitoring System and the associated reports.
Patient with a corticosteroid on repeat (and at least 3 issues in the last 12 months) will be flagged if they have not had BP, U+E, Hba1c (adults) and height (children).
Affected patients will have the ‘monitoring overdue’ exclamation mark icon:

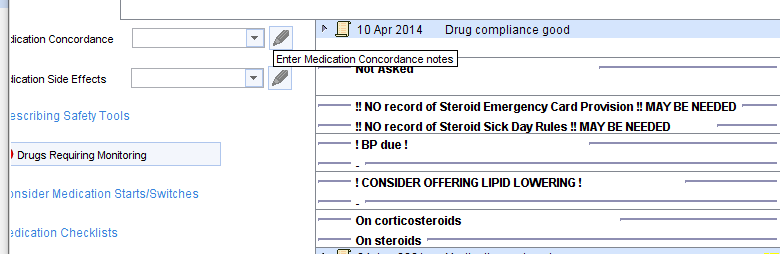
Further details will be shown on the Drugs Requiring Monitoring template (which can be launched by clicking the icon) …….
…… the Medication Review Tool template……..
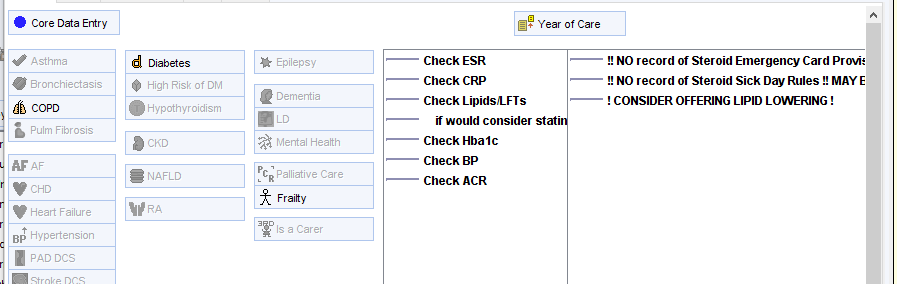
….and the LTC Master template.
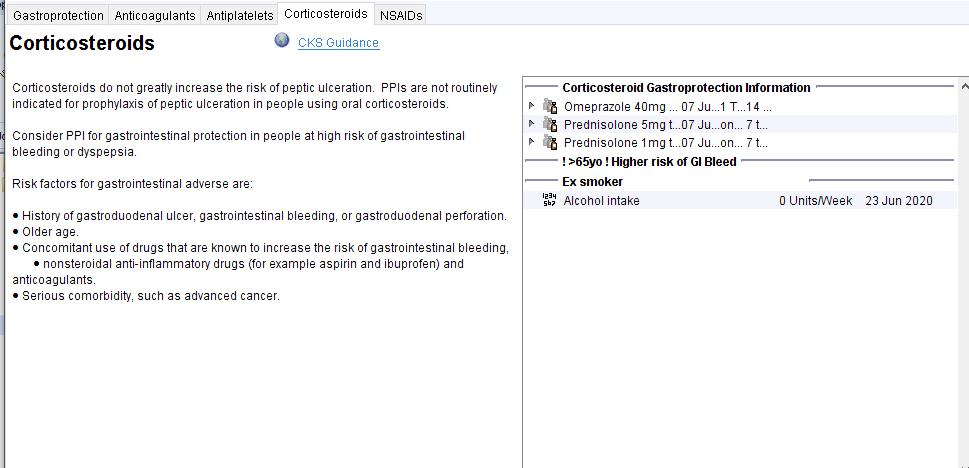
Steroid Gastroprotection
From the Steroid – CDRC template the Gastroprotection – CDRC template can be accessed. Guidance and relevant information from the record is shown (such as other drugs that can affect GI bleeding, smoking and alcohol consumption).
The following report, found in the CDRC Quality > Endocrine folder, identifies patients taking corticosteroids with higher risk of GI bleeding who are not currently prescribed gastroprotection.
Affected patients will also be flagged on the Medication Review Tool and the LTC Master templates. If a medication review if performed on the repeat template screen, the Medication Review Tool template will be opened if gastroprotection may be needed (if enabled at the unit).
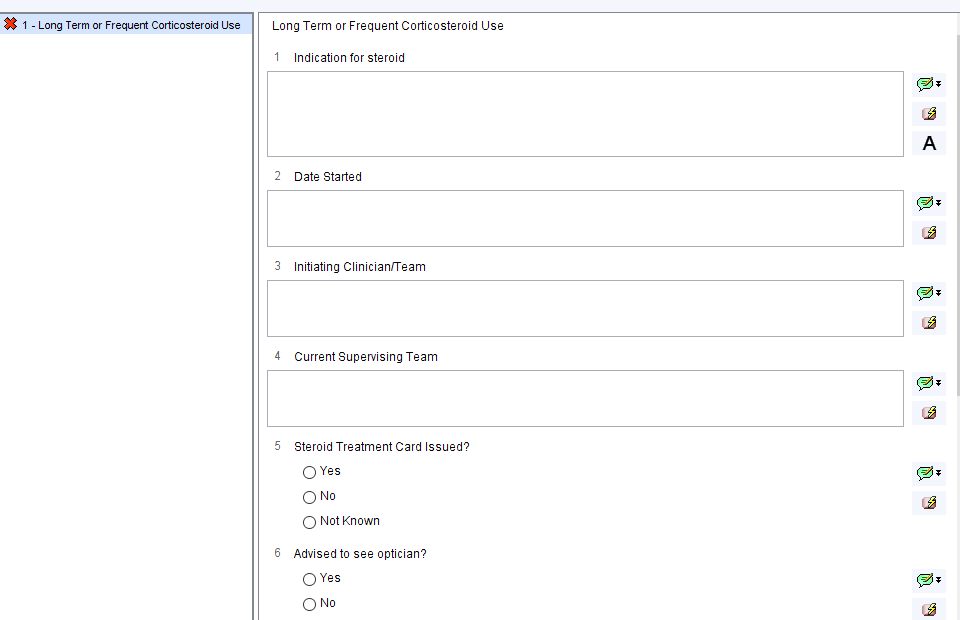
Steroid Checklist
The Steroid Checklist allows the clinician to check that appropriate information has been given to the patient and to record relevant information.
![Advised to avoid chickenpox]measles?
O Yes
O No
O Not known
Steroid Emergency Card Issued?
O Yes
O No
O Not known
Steroid Sick Day Rules Discussed PIL?
O Yes
O No
O Not Known
Finish](https://cdrc.nhs.uk/wp-content/uploads/2022/09/image-281.png)
The most recent information recorded in the checklist is shown on the Steroid – CDRC template. The checklist can be completed by clicking on the checklist button on the Steroid – CDRC template, or, if there is already an entry, right clicking on the last entry and choosing Copy Questionnaire to update the information.
