The CDRC includes a set of resources to maximise uptake of cervical screening, improve data quality and QoF performance. A systematic approach to this issue is likely to be helpful to demonstrate how quality care is being delivered, to regulators such as the CQC.
You can choose to use whichever of the elements of this system you feel will be of benefit.
- Set-up
- Data Quality/ QoF – A series of reports to improve data quality, find missing or equivocal codes, improve QoF performance and provide safety netting
- Alerts – Alerts to identify patients due for a smear
- Inviting Patients – Resources to help plan how to invite patients for smears (to augment central invitation system)
- Cervical Cancer Screening Template
Set-up
No specific setup is required for most practices using the CDRC Cervical Cytology System, although it is worth running the data quality checks and audits before using the system to minimise inappropriate alerts.
Ensure the following two Patient Status Alerts have not been disabled:

Ensure that the system-wide cervical screening recalls are being used. These are called:
Cytology Smear – for routine recall
Early Cytology Smear – for early recall
Data Quality / QoF
The CDRC Cervical Screening system has a number of reports to aid patient identification, invitation, data quality and QoF performance.
The last 3 reports will identify patients where data correction will immediately improve QoF performance.
These reports are located in the folder CDRC Quality > Gynaecology
| Report Name | Population | Recommended Action |
| * Cervical Cytology – AUDIT – 65-70 but not had smear after 50 and not declined # | Women between 65 and 70 who have not had any screening after 50. They should be offered a smear | Offer smear (could be done opportunistically or systematically) |
| * Cervical Cytology – AUDIT – Equivocal Hysterectomy Code (not business rules) | Women with a record of hysterectomy but where it isn’t clear whether or not the cervix has been removed. | Review gynaecology letters and use the No Cervix tab of the template to record a definitive hysterectomy code |
| * Cervical Cytology – AUDIT – Has cytology recall that became overdue 1m ago # | Women whose smear recall became due between one and two months ago | Can be used to supplement the Exeter invitations by identifying women who have not responded to invitation for routine or early recall |
| * Cervical Cytology – AUDIT – Has EARLY cytology recall that became overdue 1m ago # | Women whose early smear recall became due between one and two months ago | Can be used to supplement the Exeter invitations by identifying high risk women who have not responded to early recall |
| * Cervical Cytology – AUDIT – Non-Qof smear code | Women who have had a smear but this has not been recorded using a QoF code | Use the Coding tab of the template to identify the occasions when a non-QoF code was added without a QoF code. Add the appropriate QoF code |
| * Cervical Cytology – AUDIT – Recall that may need to be removed | Women who have an outstanding cytology recall that may need to be removed. – e.g. benign hysterectomy, opted out of screening | Woman has opted-out of screening – confirm that the patient has completed the opt-out form. Cancel the Cytology Recall Woman has had hysterectomy – review histology and complete the post-hysterectomy checklist. If no further smears needed, cancel the Cytology Recall. |
| * Cervical Cytology – AUDIT – Recent Result but no Future Recall (but needs recall) # | Women with a cytology result between one and two months ago who haven’t had the next recall date added or had recall suspended (e.g. due to age) | Review the record. Ensure that a plan is in place for further management e.g. next recall date, colposcopy planned, recall suspended (if informed by Exeter) |
| * Cervical Cytology – AUDIT – smear overdue (based on age or recall) | Women with screening due based on EITHER age OR overdue cytology recall | Could be used as part of a screening drive to identify patients overdue a smear (once the other reports above have been completed) |
| * Cervical Cytology – AUDIT – smear without result | Women with a smear taken code without a corresponding smear result | Review record and ensure result has returned from the laboratory and has been coded. This report works well as an Automated Reporting batch report which will run automatically at regular intervals and send a task to nursing staff to alert them to missing results. |
| * Cervical Cytology – QOF – Equivocal Hysterectomy Codes | Women due a smear (for QoF regiester) but have had an equivocal hysterectomy code | The same as the AUDIT – Equivocal Hysterectomy Code (not business rules) report above but limited to patients who are outstanding for QoF purposes |
| * Cervical Cytology – QOF – May need disclaimer code added again | Women due a smear (for QoF regiester) who have signed a screening disclaimer over 5 years ago | Check the record to make sure the patient has not changed their mind about screening. Re-add the disclaimer code using the exception tab of the template |
| * Cervical Cytology – QOF – Non QoF Smear code in last 5 years | Women due a smear (for QoF register) who have had a smear in the last 5 years using a non-QoF code | The same as the AUDIT – Non-Qof smear code report above but limited to patients who are outstanding for QoF purposes |
Patient Status Alerts
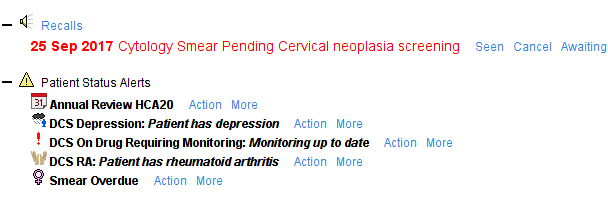
The CDRC Cervical Screening system has two alerts.
- Patients aged 25-64 – displayed if the patient has not had a smear within the expected period for her age or has an overdue Cytology Smear or Early Cytology Smear recall.
- Patients aged 65-70 – displayed if the patient has not had a smear since the age of 50
They will be displayed on the home page, under the demographics box and on the LTC Master template. This allows patients to be invited opportunistically or during annual reviews.
Either alert can be disabled if not wanted.
Clicking the female icon at the top of the screen will open the Cervical Cancer Screening Template (see below).

Inviting Patients
Patients can be invited for screening opportunistically, during LTC reviews or systematically. Alerts are displayed for the first two routes.
The following reports, found in the CDRC Quality > Gynaecology folder will allow systematic invitation:
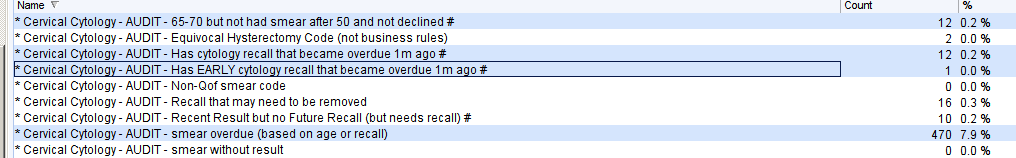
The first highlighted report identifies older women who have left the screening programme but who could still be offered a smear.
The middle two highlighted reports identify women whose smear recall became overdue between one and two months ago. These women should have already received an invitation from Exeter. These reports allow you to follow up non-responders. The Early recalls will be women who have had abnormal results and are higher risk. These searches work well if they are add to an Automated Reporting batch to send a task to the cervical screening coordinator at 28 day intervals.
The last highlighted report identifies all women who are due a smear based on their age or an outstanding cytology recall. It is worth running the data quality reports first to remove women from this report who do not need a smear e.g. benign hysterectomy, have opted out.
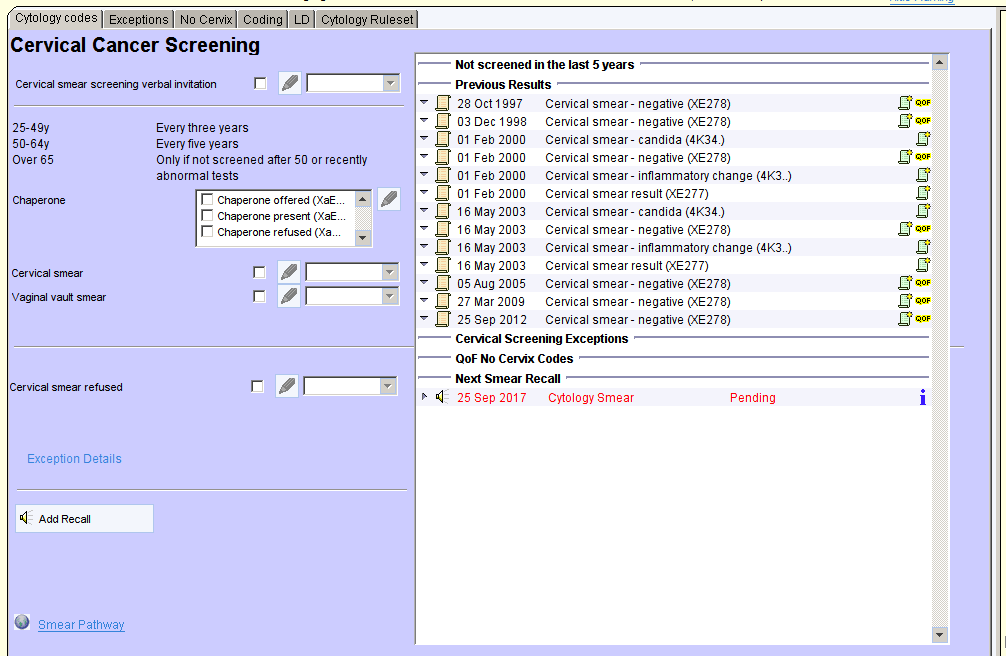
Cervical Cancer Screening – CDRC Template
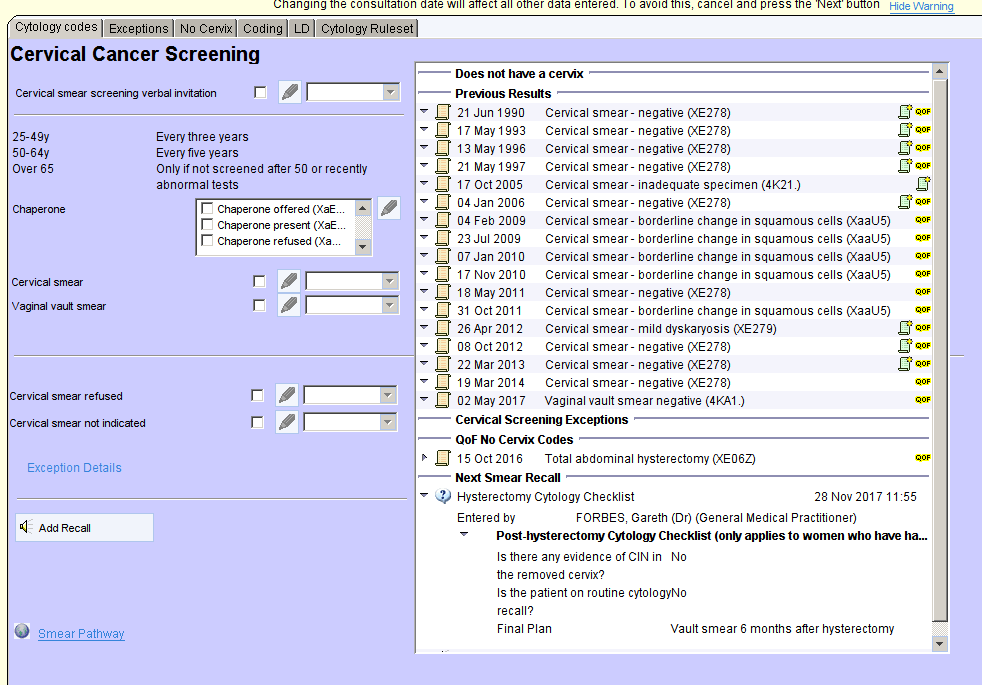
The “Cervical Cancer Screening – CDRC” template home page provides an overview of cervical screening for that patient. This includes:
- A statement to indicate if the patient has been screened within the age appropriate timescale
- Previous results (including HPV status)
- Any exception codes
- Codes indicating a lack of cervix – e.g. hysterectomy
- Details of the next recall date
- Details of any post-hysterectomy screening plan (see Hysterectomy)
The second page of the Cervical Screening Template allows entry of exception codes.
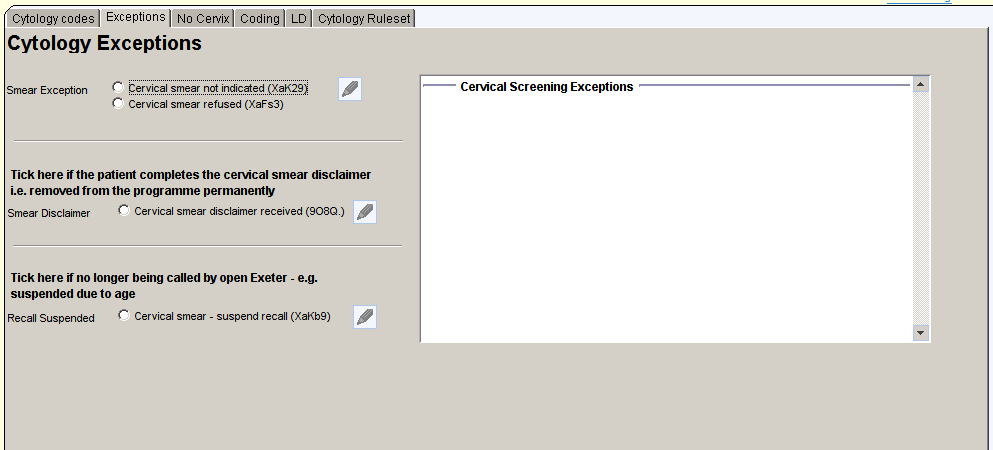
Cervical disclaimer received should only be entered when the patient has completed the official opt-out form and this has been accepted by Exeter.
Use the Recall Suspended option when Exeter have informed the practice that this is the case – this is usually due to age.
Previous exceptions are shown on the right of the template.
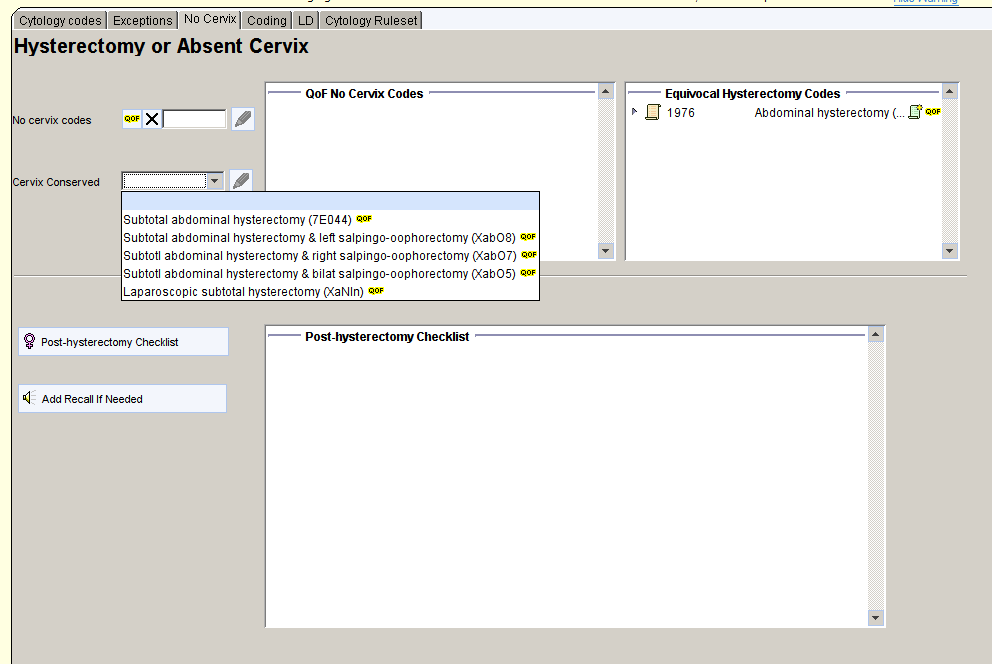
Hysterectomy or Absent Cervix
If the patient has had a hysterectomy or the cervix is absent, use the No Cervix tab.
Hysterectomies should be recorded with a code that clarifies whether the cervix has been removed or retained. The No cervix codes box provides a list of codes for hysterectomy where the cervix has been removed. The Cervix Conserved box provides a list of code for hysterectomy where the cervix has been retained.
Any equivocal hysterectomy codes (where retention/removal of cervix is not clear) are shown in the right hand table. The record should be reviewed and a definitive code added where possible.
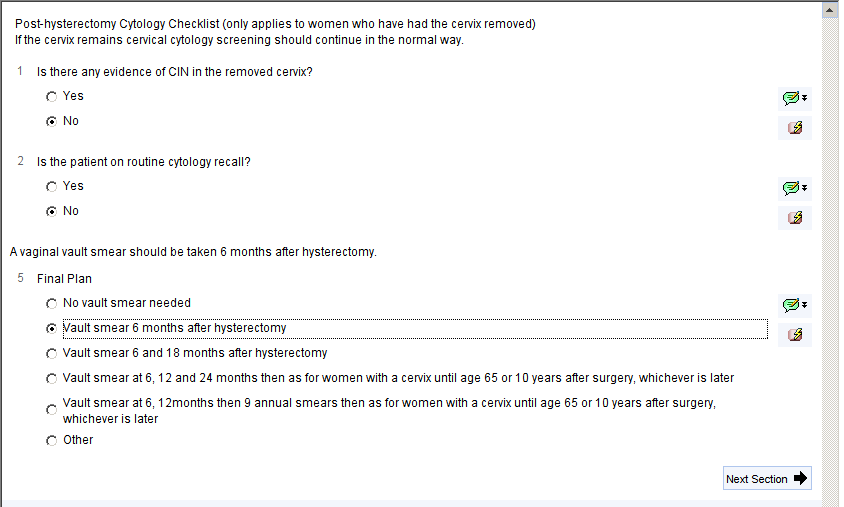
Post Hysterectomy Screening
Use the post-hysterectomy checklist to decide if further screening is needed after removal of the cervix.
A series of questions will guide you through the decision making process and display the recommended post-hysterectomy screening schedule. You should choose the appropriate option in question 5 and then set an Early Cytology Smear recall for the next smear date. This schedule will also be displayed on the cervical screening home page.
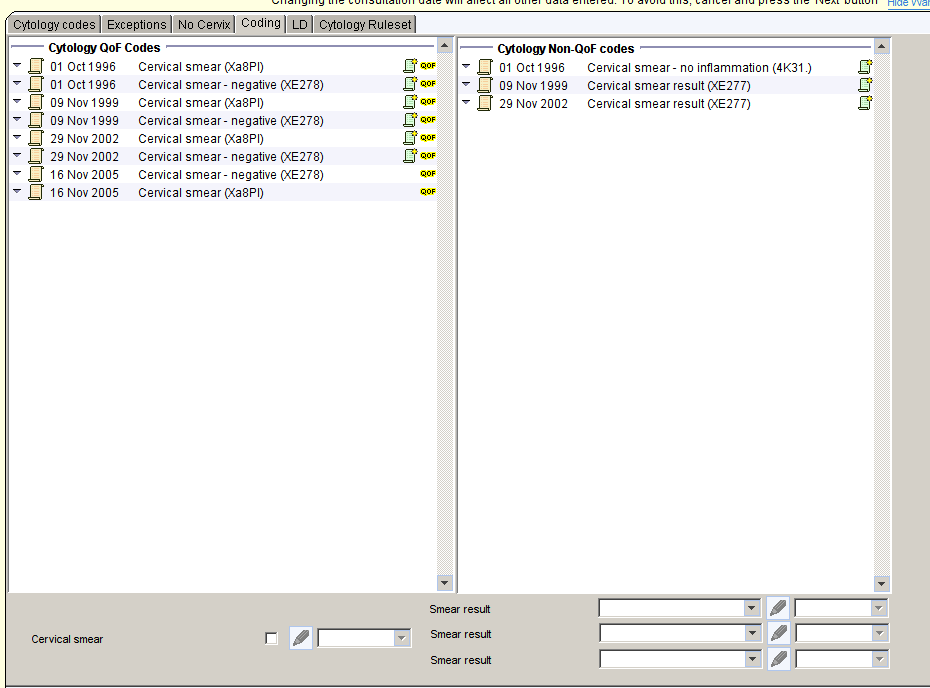
The Coding page of the template makes it easy to see if any non-QoF codes have been used to record a smear without an accompanying QoF code. The bottom of the template allows you to record the QoF code if it is missing.
The LD page has resources for patients with learning disabilities, including easy read information that can be given to or emailed to the patient/carer and best interests decision support.